Research News
Jan. 31, 2017 – Dr. Ting Chen developed a new method to accomplish efficient in vivo gene editing using ribonucleoproteins in skin stem cells of recessive dystrophic epidermolysis bullosa mouse model
Publication Date:2017/02/14
Jan. 31, 2017 – Dr. Ting Chen of NIBS, Beijing published a research article online entitled “Efficient in vivo gene editing using ribonucleoproteins in skin stem cells of recessive dystrophic epidermolysis bullosa mouse model” in Proceedings of the National Academy of Sciences of the United States of America (PNAS) (doi: 10.1073/pnas.1614775114). The paper shows that nonreplicable Cas9/sgRNA ribonucleoproteins can be used to correct genetic defects in skin stem cells of postnatal recessive dystrophic epidermolysis bullosa (RDEB) mice. This study provides a novel in vivo gene correction strategy using ribonucleoproteins as curative treatment for genetic diseases in skin and potentially in other somatic tissues.
The prokaryotic CRISPR/Cas9 system has recently emerged as a powerful tool for genome editing in mammalian cells with the potential to bring curative therapies to patients with genetic diseases. However, efficient in vivo delivery of this genome editing machinery and indeed the very feasibility of using these techniques in vivo remain challenging for most tissue types.
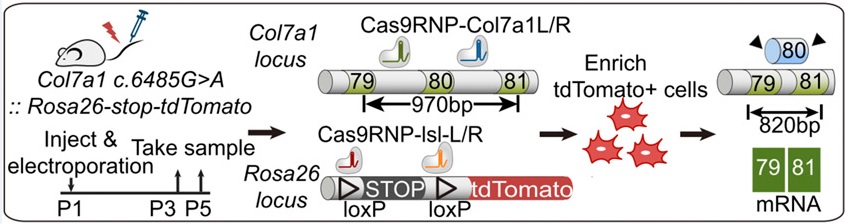
RDEB is a recessive genetic disease, caused by abnormal function of Collagen VII. In healthy skin, epidermal keratinocytes and dermal fibroblasts secrete the procollagen VII protein, which are then processed to mature collagen VII protein and it in turn forms stable homotrimers in order to assemble into network of anchoring fibrils. The anchoring fibrils are located within the basement membrane zone (BMZ) between the epidermis and dermis, where they participate in stabilizing the association of the epidermis to the underlying dermis. In recessive dystrophic epidermolysis bullosa (RDEB) patients, mutations in the Col7a1 gene cause absent or dysfunctional collagen VII protein production, which leads to defective epidermal-dermal adhesion.
Here, the authors demonstrated the in vivo use of Cas9/sgRNA ribonucleoproteins to mediate gene correction in skin stem cells of postnatal RDEB mice. First the authors generated a novel RDEB mouse model based on a patient specific point mutation and proved that the mouse shows RDEB hallmarks. Then they established a mouse model missing the exon containing the point mutation in Col7a1 gene to prove the efficiency and safety of exon skipping as a gene correction method. To apply the gene correction system in postnatal skin, the authors developed a method to deliver nonreplicable protein/RNA complexes in vivo via electroporation; it induced one-time, permanent modification of genomic DNA in skin stem cells. Finally they demonstrated the feasibility and efficiency of curative gene editing system to permanently restore the function of the collagen VII protein in vivo. Owing to the efficiency and simplicity of this method, the authors have set the stage for the use of ribonucleoproteins for a broad spectrum of genetic diseases in somatic tissues beyond skin.
The PTN program Ph.D candidate student Wenbo Wu in the Ting Chen’s lab is the first author. Other contributing authors include Zhiwei Lu, Fei Li, Wenjie Wang, Nannan Qian from the Ting Chen’s lab, Jinzhi Duan and Dr. Yu Zhang from Yu Zhang’s Lab as well as Dr. Fengchao Wang from Transgenic Animal Center. Dr. Ting Chen is the corresponding author. This research was supported by a grant of the Beijing Municipal Science & Technology Commission (Z151100003915080) and the grant of the 973 Program. This work was carried out at the National Institute of Biological Sciences, Beijing.